Do you have some time to travel? What about UK? Some nice places to be
November 2025 regulatory highlights: EU Deforestation Regulation update, QMS and ISO standards, EUDAMED changes, and a look at UK destinations worth visiting.
11/5/20254 min read


In this busy month of November, with final preparations underway for strong sales in 2025, we keep a compact watch on regulatory developments that matter to economic operators — without losing sight of the human side: a few travel suggestions in the UK to inspire your next short break. Written by Tamara Fayolle, ADDE Regulatory Affairs Department.
To reduce the load that the IT system of the EUDR will need and also more time to economic actors: a six-month postponement for micro and small enterprises to 30 December 2026 (from 30 June 2026) , as well as for the newly defined category of micro and small primary operators, while for large and medium (non-SME) operators and traders, it will apply as originally planned from 30 December 2025 onward.


But to ensure a gradual phase-in of the rules, these large and medium sized actors will benefit from a grace period of six months for checks and enforcement.
You sell for example latex gloves or dental cupules of rubber? You are concerned.
The European Parliament and the Council would need to formally adopt the amendment before it can come into effect.
EU Deforestation Regulation
Guidelines – QMS requirements – Measurements and analyses
| What?
The guidelines (for example MEDDEV, MDCG) are not legally binding. However, due to the participation of the interested parties and the experts from competent authorities, it is expected that the guidelines be followed, will ensure the uniform application of relevant directive and regulation provisions.
| Why?
All kind of subjects. The question today is Are you up to date with the latest medical device QMS requirements?


Importers and distributor can become compliant with ISO 13485 on a volontary basis or you found out you repackage medical devices or consmetics and thus become a regulatory manufacturer.
Anyway, make sure you have process for monitoring and measuring customer satisfaction and that you collect, analyze and act on your customer feedback and inquiries.
You have to handle and investigate complaints even if the manufacturer is primarily concerned. Although you are not required to draw conclusions on behalf of manufacturers, you must actively participate in identifying claims and forward them without delay.
Comment or suggest
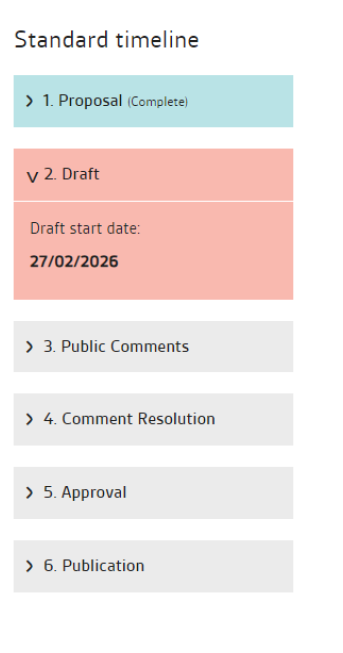
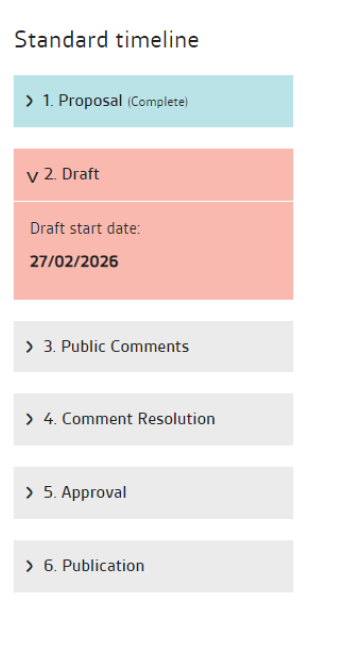
The standards development process is divided into chronological stages: Proposal; Drafting; Public comment; Comment resolution; Approval; Published standard
BS EN ISO 6877:2025 Published standard begun: 2025-08-14 Endodontic obturating materials.
PD ISO/TS 4640 Test methods for tensile bond strength to tooth structure Drafting begins: 2026-02-27.
BS EN ISO 9917-2 Water-based cements. Part 2: Resin-modified cements Drafting begins: 2026-06-26.
You can comment on proposed and draft standards or suggest an idea for a new standard.
Source: Committee TC 106
Dentistry - Drafts
EUDAMED - Consult the Database
Ensure your medical devices meet EU regulations
The new version of EUDAMED 2.18.0 has been deployed October 2025
You are right Distributors are not registrated but you can get a lot of information of the products you daily sell. an additional tool so that you can verify the compliance.
Detect changes to reduce risks – Mouthwashes
Build your library
Sometimes it may be unclear whether a particular product is a cosmetic product under cosmetics legislation or whether it falls under other sectorial legislation.
Collect the guidance documents.
Mouthwashes and dental gels
Is a mouthwash or a dental gel (other than regular toothpaste) a cosmetic product?
Mouthwashes and dental gels may be considered as cosmetic products provided that they are intended to be placed in contact with the teeth and the mucous membranes of the oral cavity and the purpose of their use is “exclusively or mainly cleaning them, perfuming them, changing their appearance, protecting them, keeping them in good condition or correcting body odours”.
Mouthwashes and dental gels with secondary "antimicrobial" claims may be considered as cosmetic products provided that the main purpose of use is that of a cosmetic product.
A product which is presented or intended for the treatment or prevention of infections, inflammation or other oral cavity diseases should not be considered as a cosmetic product.
In any case, a decision on the qualification of the products has to be made by the national competent authorities, on a case-by-case basis, and taking into account all the characteristics of the product, such as the presentation of the products, the ingredients, the mode of action and instructions of use.
Source: MANUAL OF THE WORKING GROUP ON COSMETIC PRODUCTS (SUB-GROUP ON BORDERLINE PRODUCTS) ON THE SCOPE OF APPLICATION OF THE COSMETICS REGULATION (EC) NO 1223/2009 (ART. 2(1)(A)) Version 5.5 (JUNE 2025)
You are maybe also concerned by the
COMMISSION IMPLEMENTING DECISION (EU) 2025/2078 of 17 October 2025 amending Implementing Decision (EU) 2021/1182 as regards harmonised standards for surgical clothing and drapes, medical face masks and sterilizers for medical purposes. EN ISO 13795-1:2025, EN ISO 13795-2:2025, EN ISO 14683:2025 et EN ISO 14180:2025 – are updated.
© 2026 ADDE – Association of Dental Distributors in Europe. All rights reserved.






